Titratable Acidity, pH, and Buffering
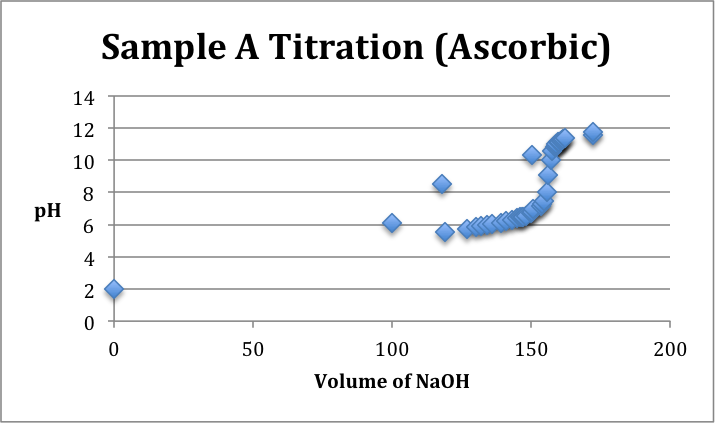
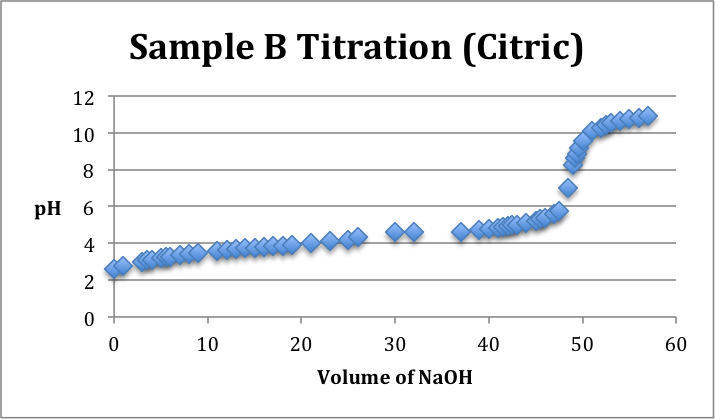
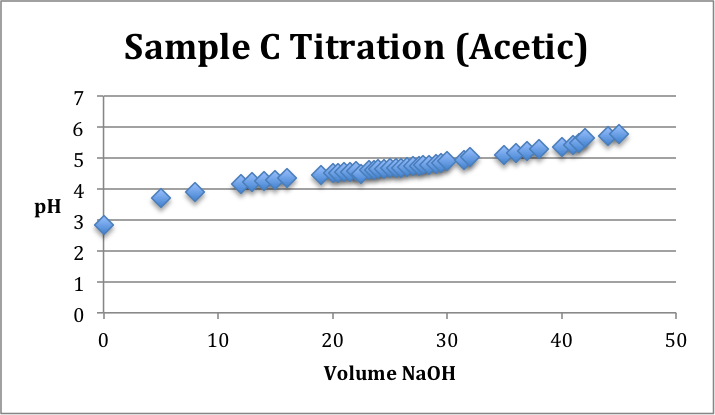
Sample Identification: Unknown samples A, B, and C. Sample A was identified as Ascorbic Acid. Sample B was identified as Citric Acid. Sample C was identified as Acetic Acid.
Purpose: The purpose in this experiment was to identify unknown acids through the measurement of acidity via pH meter and titration
Purpose: The purpose in this experiment was to identify unknown acids through the measurement of acidity via pH meter and titration
Conclusion/Comments: Sample A was identified to be Ascorbic Acid, Sample B was identified as Citric Acid, and Sample C was identified as Acetic Acid. To make these conclusions it was noted that pH=pKa at the half equivalence point. Ultimately, we were trying to match the curves with the corresponding number of pKa values given in the lab protocol. Acetic Acid had one pKa value, Ascorbic Acid had two pKa values, and Citric Acid had three pKa values. Our data, as depicted on the titration curves above, was not the best. We had to do our best with the data that we had to identify which half equivalence point pH’s corresponded the best with their appropriate pKa values from the protocol. To improve upon our results in the future, it is important to acknowledge that time is of the essence. It was difficult to finish so efficiency in the future is key. Additionally, it may be better to write out and record more data points to get a more representative curve that actually displays equivalence points on the graphs. Titrations are very sensitive and therefore results can fluctuate without warning.