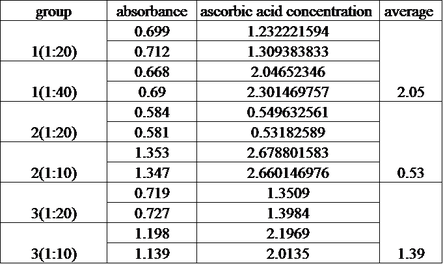Background: Titration
Objective
To determine the concentration of ascorbic acid in the fruit juice by the use of spectrophotometer and compared with titration.
Result
Objective
To determine the concentration of ascorbic acid in the fruit juice by the use of spectrophotometer and compared with titration.
Result
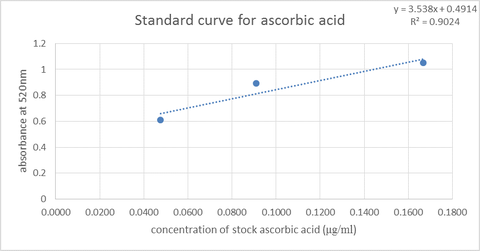
Ascorbic acid concentration (μg/ml)
Refrigeration: 2.05*200 = 410
Room temperature: 0.53*200 =106
Unknown: 1.39*200 = 278
The nutrition label: 450
Comments
The ascorbic acid in three sample is: refrigeration > unknown > room temperature. The orange juice that stored in the fridge is most close to the nutrition label given. It demonstrates that temperature plays an important role in ascorbic acid storage. From the result, if the unknown sample is stored for similar days as other two samples, we can predict that the unknown sample is store at temperature range from 4oC to 25oC.
Limitation
We noticed that the sample concentration in different dilution varies a lot, the possible reasons for this are:
1. Systematic error: the accuracy of spectrophotometer or the fluctuation of spectrophotometer
2. Random error: the real concentration of sample is not as the same as its dilution, due to the random error occurs in every stage.
3. The homogeneous: the juice sample is not exactly homogenous, which decreases the validity.
4. The ascorbic acid loss during each stage: ascorbic acid is easy to degrade at room temperature, so all the sample should be measure as soon as possible. In addition, the test tubes should be capped each time after used, in order to reduce ascorbic acid contact with oxygen.
Modification
In order to improve this experiment
1. Vortex samples thoroughly or centrifuge samples
2. Make more series of dilution for sample
3. Improve the accuracy of standard curve by collecting more data (eg: more dilutions
Interested in Vitamins? What to learn more?
Refrigeration: 2.05*200 = 410
Room temperature: 0.53*200 =106
Unknown: 1.39*200 = 278
The nutrition label: 450
Comments
The ascorbic acid in three sample is: refrigeration > unknown > room temperature. The orange juice that stored in the fridge is most close to the nutrition label given. It demonstrates that temperature plays an important role in ascorbic acid storage. From the result, if the unknown sample is stored for similar days as other two samples, we can predict that the unknown sample is store at temperature range from 4oC to 25oC.
Limitation
We noticed that the sample concentration in different dilution varies a lot, the possible reasons for this are:
1. Systematic error: the accuracy of spectrophotometer or the fluctuation of spectrophotometer
2. Random error: the real concentration of sample is not as the same as its dilution, due to the random error occurs in every stage.
3. The homogeneous: the juice sample is not exactly homogenous, which decreases the validity.
4. The ascorbic acid loss during each stage: ascorbic acid is easy to degrade at room temperature, so all the sample should be measure as soon as possible. In addition, the test tubes should be capped each time after used, in order to reduce ascorbic acid contact with oxygen.
Modification
In order to improve this experiment
1. Vortex samples thoroughly or centrifuge samples
2. Make more series of dilution for sample
3. Improve the accuracy of standard curve by collecting more data (eg: more dilutions
Interested in Vitamins? What to learn more?
- Fortify your knowledge about vitamins by FDA: http://www.fda.gov/ForConsumers/ConsumerUpdates/ucm118079.htm
- And a video from FDA